ANANDA Scientific Announces FDA approval of the IND for the Clinical Trial on the Treatment of Opioid Use Disorder (OUD)
Por um escritor misterioso
Descrição
ANANDA Scientific Inc., (a biotech pharma company) today announced approval by the U.S. Food and Drug Administration (FDA) of the Investigational New

Google Pilots Opioid Addiction Moonshot in Dayton, Ohio - Bloomberg

FDA-approved medications for OUD, with typical dosing paradigms for
Articles about Ananda Scientific

g556911.jpg
4 Medications for Opioid Use Disorder in Various Treatment Settings, Medications for Opioid Use Disorder Save Lives

ANANDA Scientific and David Geffen School of Medicine UCLA Announce Clinical Trial Utilizing Liquid StructureTM Cannabidiol (CBD) for the Treatment of Opioid Use Disorder (OUD)

A Decade of FDA-Approved Drugs (2010–2019): Trends and Future Directions

ANANDA Scientific Announces FDA approval of the IND for the Clinical Trial on the Treatment of Opioid Use Disorder (OUD)
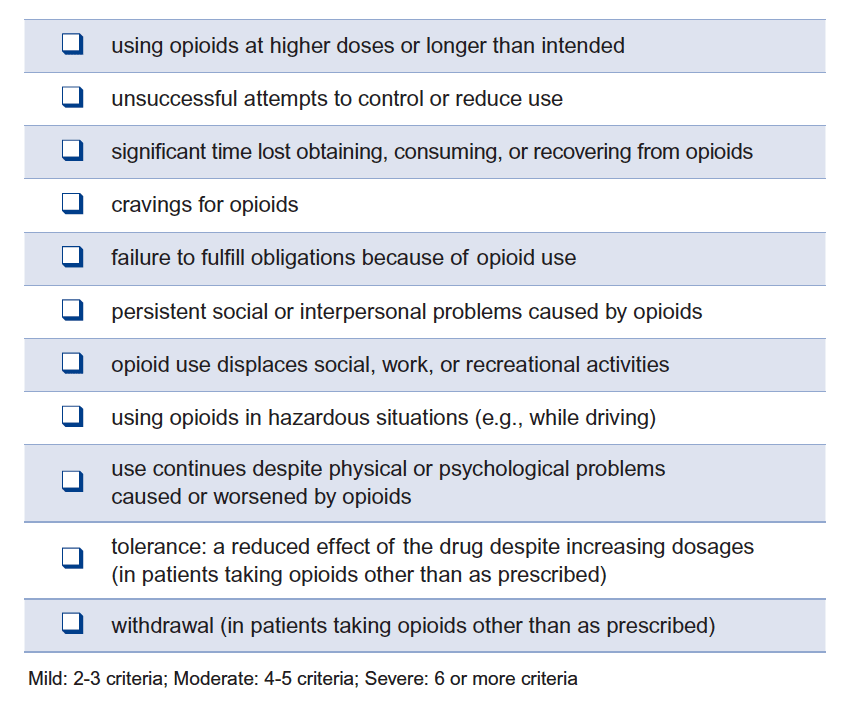
Opiod Use Disorder - OUD Clinical Module

FDA-Approved Medications for the Treatment of OUD

Issue Brief: Treating Opioid Use Disorder in Justice-Involved Populations - Justice Community Opioid Innovation Network Coordination and Translation Center
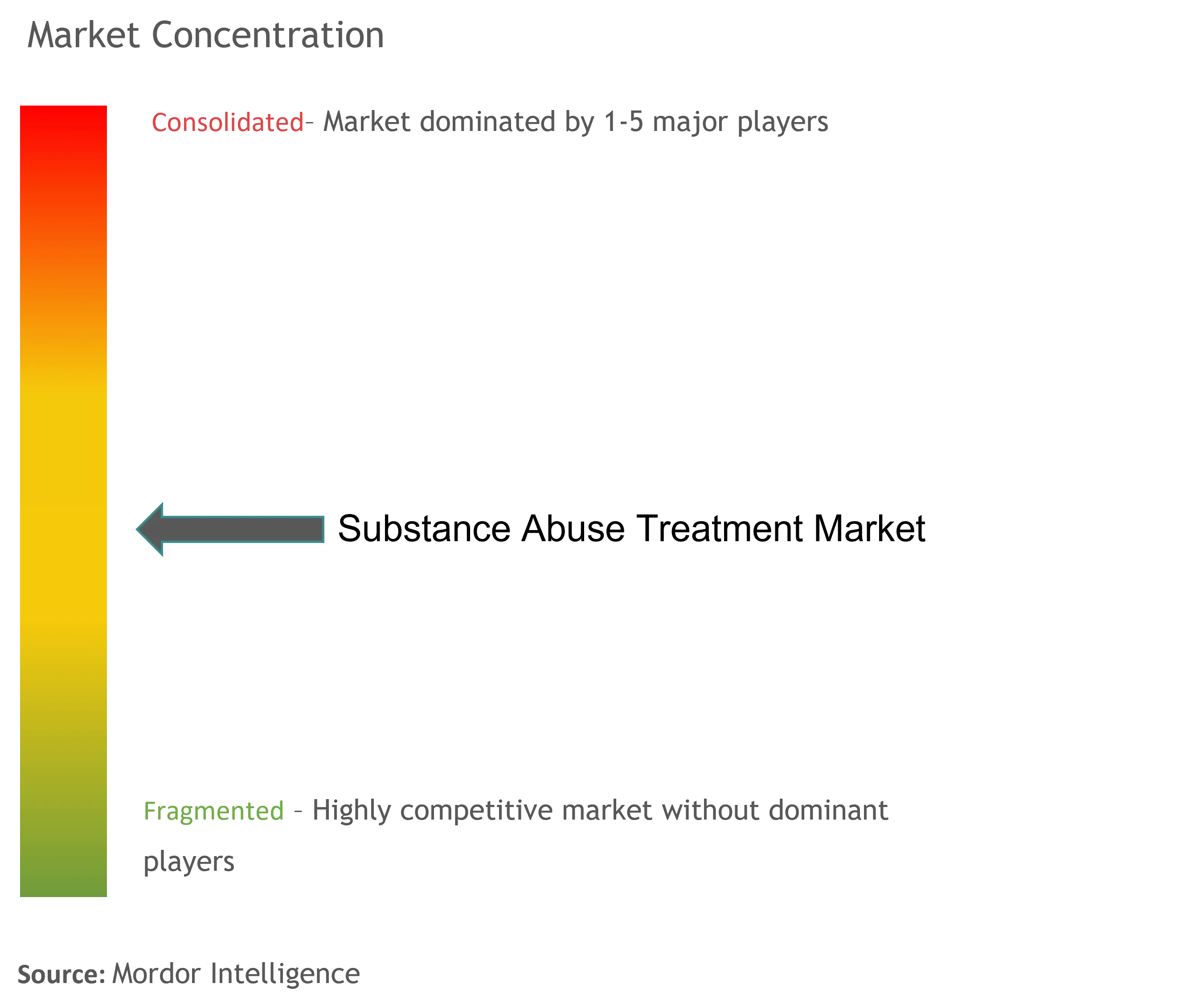
Substance Abuse Treatment Market Size & Share Analysis - Industry Research Report - Growth Trends

The Opioid Crisis and Recent Federal Policy Responses
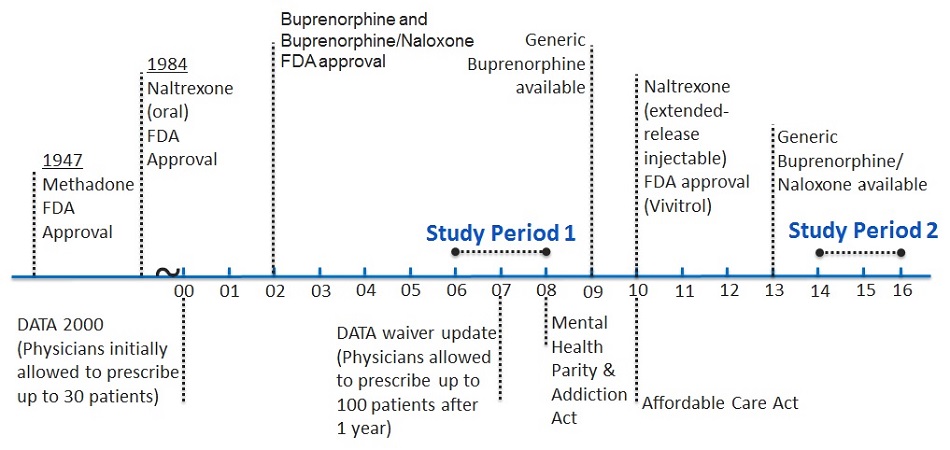
Use of Medication-Assisted Treatment for Opioid Use Disorders in Employer-Sponsored Health Insurance: Final Report

Blog Articles And Insights
de
por adulto (o preço varia de acordo com o tamanho do grupo)







