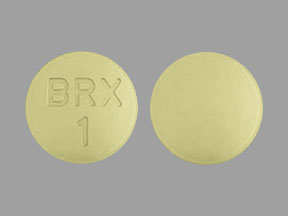
Otsuka and Lundbeck Announce U.S. Food and Drug Administration (FDA) Approval of Supplemental New Drug Application (sNDA) for REXULTI® (brexpiprazole) for the Treatment of Agitation Associated with Dementia Due to Alzheimer's Disease
Por um escritor misterioso
Descrição

Brexpiprazole Gets Priority Review for Agitation With Alzheimer

FDA Accepts sNDA for Rexulti for Alzheimer's Agitation

The Alzheimer's drug lecanemab wins full FDA approval. It's a very

Mischa Agster on LinkedIn: Otsuka Announces New FDA Approval

Otsuka and Lundbeck Issue Statement on U.S. Food and Drug

FDA - Denmark

Allison Rosenthal on LinkedIn: So proud and humbled to announce a

Positive Phase III Alzheimer's results send Lundbeck and Otsuka

Otsuka and Lundbeck gain FDA approval for supplemental New Drug

Alzheimer's - Page 2 of 4 - Drug Discovery World (DDW)
Articles about Lundbeck

Lundbeck, Otsuka drug cut agitation in Alzheimer's patients

Allison Rosenthal على LinkedIn: National Family Caregivers Month

Brian Ruhl on LinkedIn: The Neurological Benefits of Building Trust

John J. Miller, M.D. en LinkedIn: Yes, It's All in Your Head
de
por adulto (o preço varia de acordo com o tamanho do grupo)